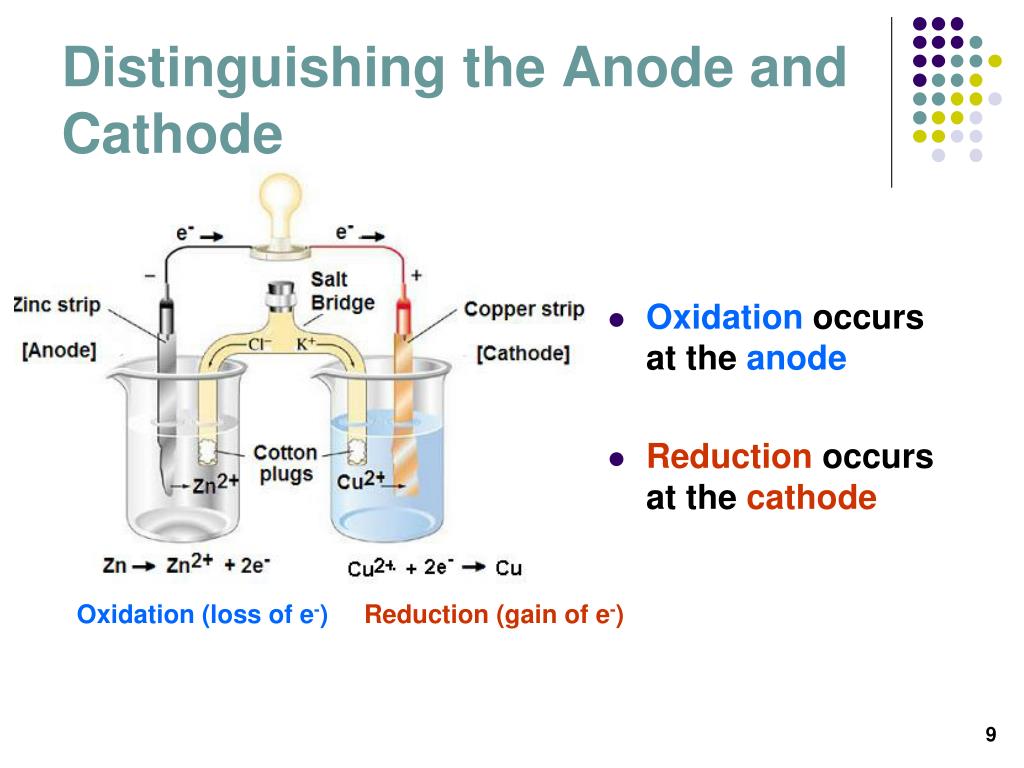
Before electrolysis, two electrodes are dipped in the solution of NaCl. However, both cations and anions have opposite roles in general.įor a better understanding of the electrolysis of a cathode and anode, Let’s take the example of the electrolysis of molten NaCl. As far as the charge of ions is concerned in an electrolytic cell, an anion is a positively charged, while a cation is a negatively charged ion. An electrolytic cell is a device that changes electrical energy into chemical energy. During the process of electrolysis in a chemical reaction, the change occurs through electricity transmission in the circuit. You can study the electric movement of any solution or suspension using a cathode or anode including aqueous solutions. This means that the charge of the cathode will turn into a positively charged electrode. Whereas the cathode can act as an anode in a galvanic cell. It means that the charge of the anode will turn into a negatively charged electrode. In a galvanic cell, the anode can act as a cathode. CathodeĬonversely, a reduction reaction occurs at a cathode electrode in an electrolytic cell. The anode is the location for oxidation reaction in the electrolytic cell. CathodeĪt the same time, the cathode typically acts as a negatively charged electrode. The anode is mostly on the positive side, depending on the type of cell used during the reaction. CathodeĬontrarily, in the cathode, electricity flows outward. In the anode, electricity flows inside the device. On the other hand, the cathode is the negative electrode in an electrolytic cell that releases electrons. The anode is the positive electrode in an electrolytic cell that acquires electrons. On the contrary, cathode translates to “way downwards” in its origin word. Easy in terms of fabrication Difference Between Anode and Cathode Literal Meaning AnodeĪnode comes from a Greek word that means “way upwards.” Cathode.Remains in control when it comes in contact with an electrolyte.Cathode works as an efficient, reducing agent. 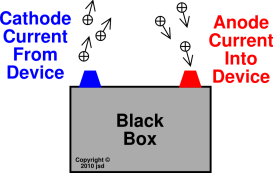
These tubes are used in vacuum tubes, discharge tubes, discharge lamps, etc. In comparison, the filament does not heat the cold cathodes. Hot cathodes are heated by electric current passing through the filament. A cathode electrode is of two types a hot cathode and a cold cathode. In a galvanic cell, a cathode is a copper metal dipped in a CuSO 4 solution. Cathodes are often used in electron microscopes. The word “cathode” originated from a Greek word that means “way towards downwards.” Generally, a cathode is an electrode where reduction takes place, which means that the cathode gains electrons from the external circuit and gets reduced. They are low-cost electrodes What is the Cathode?Ī cathode is a negatively charged electron that allows electrons to enter an electric device.Anodes have a good level of electrical conductivity.Anodes work as an efficient oxidizing agent.Anodes act as positive electrodes in electrolytic cells and negative electrodes in galvanic cells.Anode and cathode also help understand the electric current in other liquids and aqueous solutions. The anode is a zinc metal in a galvanic cell dipped in a ZnSO 4 solution. In contrast, in a galvanic cell, the anode acts as a negative electrode as it has a negative ability compared to a galvanic cell’s solution. An anode acts as a positive electrode in an electrochemical reaction because electrochemical reactions use electrical energy to transmit a chemical reaction. In simple words, an anode is a point where an oxidation reaction takes place. “Anode” is a Greek word that means “way upwards.” Although, anodes are positively charged electrodes in electrolytic cells, they act as a negative electrode in a galvanic cell. The anode is the positively charged electrode of an electrochemical cell that attracts electrons. Galvanic Cell Negative Positive What is an Anode? The Bottom Line Comparison Table Factors Anode Cathode Meaning Way upwards Way downwards Definition Positive electrode Negative electrode Electricity Flow Inwards Outwards Associated Rxn.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
