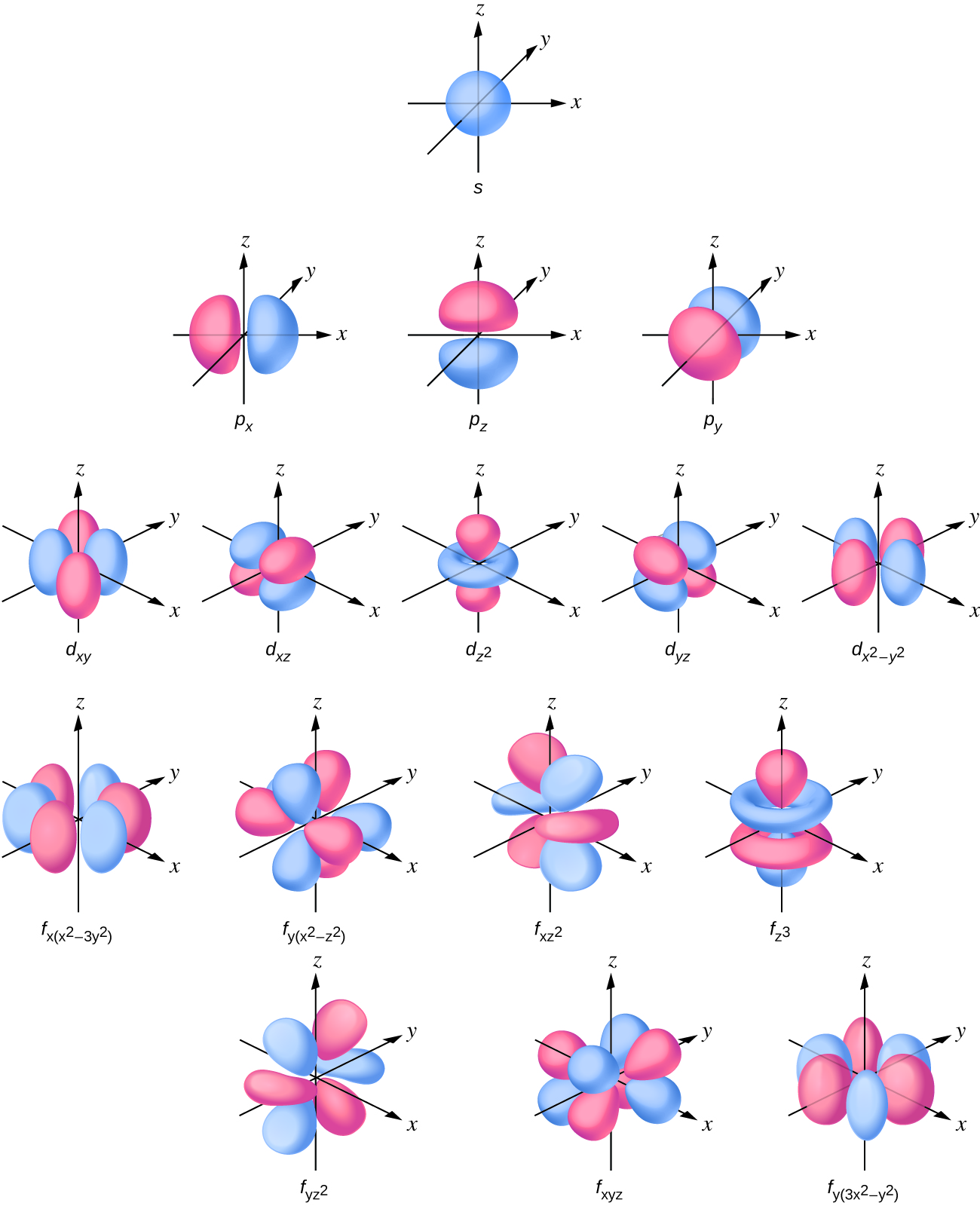
Solutions of the atomic Schrödinger equationĪs stated in the introduction, the original meaning of atomic orbital is the quadratically integrable eigenfunction of a one-electron atomic Schrödinger equation. Solid harmonics depend on the spherical polar coordinates of r A1. 
It is common to choose solid harmonic functions (which differ from spherical harmonics only by a factor r A1 ℓ, the ℓth power of r A1) as the angular parts of the one-electron basis functions. Functions of r A1 (the distance of electron 1 to nucleus A) are used as the radial parts of basis functions depending on r A1, the position vector of electron 1 with respect to a system of axes with origin on A. One-electron basis functions (briefly orbitals) are needed in many numerical methods for solving the Schrödinger equation of atoms and molecules. For such functions it is meaningless to speak of their (orbital) energy. Later in the development of quantum chemistry, especially in its computationally oriented branches, the term "atomic orbital" simply became to mean a one-electron function centered on an atom, not necessarily the solution of an effective one-electron atomic Schrödinger equation. The latter functions determine the shape of the AOs. If, in addition, the effective Schrödinger equation is invariant under rotation, like the Schrödinger equation of the one-electron atoms, it can be shown that the angular parts of the solutions consist of spherical harmonic functions. Because the effective equation depends only on the coordinates x, y, z of the single electron that is considered, the solution (eigenfunction) is a function of x, y, and z-an atomic orbital. A well-known mean-field method is the one proposed by Hartree and Fock. It must be replaced by an effective one-electron equation that contains the interaction between the electrons in an averaged manner a single electron is considered that moves in a mean-field. 
In the case of more-electron atoms the exact Schrödinger equation is not solvable. In the case of one-electron atoms the Schrödinger equation is exactly solvable, that is, its solutions are known in analytic form. In the pre-computer era of quantum chemistry, an atomic orbital (AO) was seen as a solution of a one-electron Schrödinger eigenvalue equation for an atomic electron. In quantum mechanics and quantum chemistry, an atomic orbital is a function of an electron that describes the motion-in the quantum mechanical sense of the word-of the electron around the nucleus of an atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
